During the last decades, advances in diagnostic imaging techniques and in surgical solutions led to the need for developing computer tools to support surgical procedures planning, through the assessment of preoperative measurements, and prediction of postoperative outcomes.
Particularly, knowledge of the Mitral Valve (MV) anatomy still represents a challenging task owing to its highly complex geometry, inter-patient variability of valvular dimensions, poor and discordant data reported in literature. None of the existing 3D MV models fulfill the mandatory conditions to be compatible with everyday clinical practice: patient-specific and parametric models based on data available from routine exams, almost completely automated, fast and user-friendly software are increasingly demanded to clearly visualize anatomical structures, to understand physiology of valvular pathologies and to select candidates for specific therapies [1].
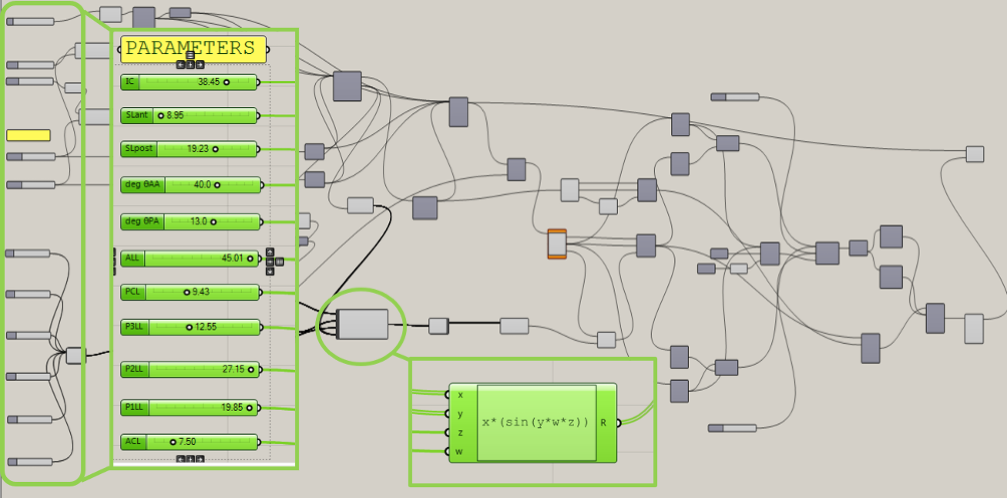
During her research activities, our CompMech Group member, Laura Mazzocchi, directly interacted with medical operators routinely performing heart valves ultrasound evaluations, Dr. Fabiana Gamabrin and Dr. Giulia Magrini, cardiologists at San Pio X Clinic in Milan and San Matteo Hospital in Pavia, respectively. She identified a series of anatomical values necessary to the implementation of a three-dimensional parametric model of the Mitral Valve. This algorithm allows to quickly generate models depending on different entered values of parameters measured during 3D echocardiographic exams and to modify valve geometry in real-time, as shown in the following video.
To realize this model, it has been used a highly versatile commercial CAD software, Rhinoceros, which is an accurate free-form surface modeling tool that allows to create almost any 3D object. Many plug-ins are available to expand its capabilities: among them, the graphical algorithm editor Grasshopper. Its layout is kept to minimal, but it allows to create complex geometries and solutions, very difficult to construct otherwise. Its strength is the possibility to edit the model real-time, to change and update original parameters and to refine the rules definition while working. The output parameters of each component represent the input for the subsequent one, thus changes of a parameter values will be spread over the whole model (Fig. 1).
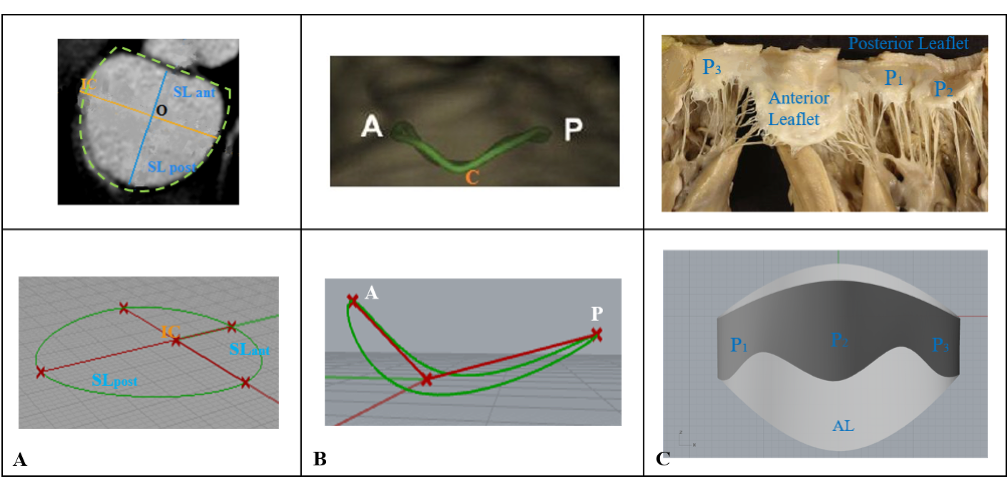
The reconstructed geometrical model shows macroscopic anatomic features of the Mitral Valve in diastolic phase. The incorporated valvular substructures are the saddle-shaped annulus, the anterior leaflet consisting of a single cusp, and the tri-scalloped posterior leaflet. Fig. 2 depicts the main steps of model generation, compared with real valves images.
Fig. 2: Steps for geometrical reconstruction of Mitral Valve parametric model. The D-shape annulus is obtained through the union of two semi-ellipses, defined from two main annular diameters. The anatomical saddle-shape is obtained by a septo-lateral rotation. The leaflets free edge profile is described by leaflets height maximum value in the middle of each scallop.
References
[1] Stevanella M., et al. “Mitral Valve Finite Element Modeling: Implications of Tissues’ Nonlinear Response and Annular Motion”. J Biomech Eng-T ASME, 2009; 131: 121010-1-9.
May 15th, 2020